
Life Among the Dead
Biodiversity (and Venom) Among Millions of Insect Specimens
On November 4, 1909, at a banana farm in eastern Costa Rica, Philip Powell Calvert, a legend in American entomology, swung his insect net and landed a dragonfly with clear wings and a golden body. As he would innumerable times at places around the planet, Calvert dispatched the insect, placed it in a folded triangle of paper, noted the date and location and then went on swinging for more.
 But this wasn’t just any dragonfly. It was one of the most adventuresome animals on Earth, a ruler of air and water, a predator and opportunist, but more than anything a world traveller able to cross oceans and exploit new lands with the tenacity of conquistadors. Calvert had caught a Pantala flavescens (Wandering Glider).
But this wasn’t just any dragonfly. It was one of the most adventuresome animals on Earth, a ruler of air and water, a predator and opportunist, but more than anything a world traveller able to cross oceans and exploit new lands with the tenacity of conquistadors. Calvert had caught a Pantala flavescens (Wandering Glider).
More than a century later, here in Gainesville, Florida, I’m hot on Calvert’s trail, holding his dragonfly specimen with the humility due any artifact, as if this dried insect were one of Darwin’s finches or a page from Linnaeus’s notes. The treasures here before me are vast. I am among one of the world’s great scientific collections of dragonflies — more than 600,000 specimens housed at the Florida State Collection of Arthropods (FSCA), which itself holds an estimated 9 million specimens from nearly every corner of the planet.
For three weeks in January (during a brutal Polar Vortex at home in Vermont) I floated in a warm whirlwind of dead insects.
Mostly I drifted among 1,312 specimens of Pantala, about which I’m writing a book. From New Mexico to New Guinea, Turkey to Trinidad, Cuba to China, in this collection are Pantala specimens collected from no fewer than 59 nations and 30 U.S. states. These specimens, including Calvert’s, which now reside in clear mylar envelopes and are organized like a massive card catalog, represent nothing short of the distant journeys of a worldly insect.
Pantala has been netted on every continent except Antarctica. (Give it time.) Seated with a drawer full of them, I was trailing intrepid biologists who had chased this dragonfly for me around the world for more than a century. They went places I will never go. But their journeys are now mine. And their insects still tell stories no less monumental than the rise of life on Earth and how it all fits together. But only if we care to look.
Although the classical era of biological collecting is in many respects now over, new technologies that Darwin and Linnaeus could never have envisioned are bringing to life dried specimens that have resided on pins in dusty boxes for centuries. Among these dead we can discover ways to save the living.
Calvert’s dragonfly from Costa Rica is but one example. A world traveller, Pantala species migrates like birds on the winds. Its common name, depending on where you find it, is (in the Americas) Wandering Glider and (in Europe) Wandering Globetrotter. So from where might Calvert’s dragonfly have wandered or glided?
We know, from Calvert’s notes, that he caught it among bananas growing in Costa Rica’s eastern Limon Province. Beyond that, its provenance Calvert could not have known. And that’s okay. A century ago we were still learning about the inclinations of wild animals. So discoveries anywhere were illuminating. Yet these specimens still have stories to reveal. And only now are we able to discover the flight paths of long-dead wildlife. The dragonflies themselves still carry their roadmaps. All you need in order to know the route is a bit of basic atomic physics.
Going Nuclear
It turns out that we can catch a dragonfly, or even take one from a museum collection, analyze its tissue and determine roughly how far it has flown. Our tracer is water, H2O, or more to the point those two hydrogen atoms. And here is where one minute of reading about atomic physics can help you understand hours and hours of new research in wildlife biology.
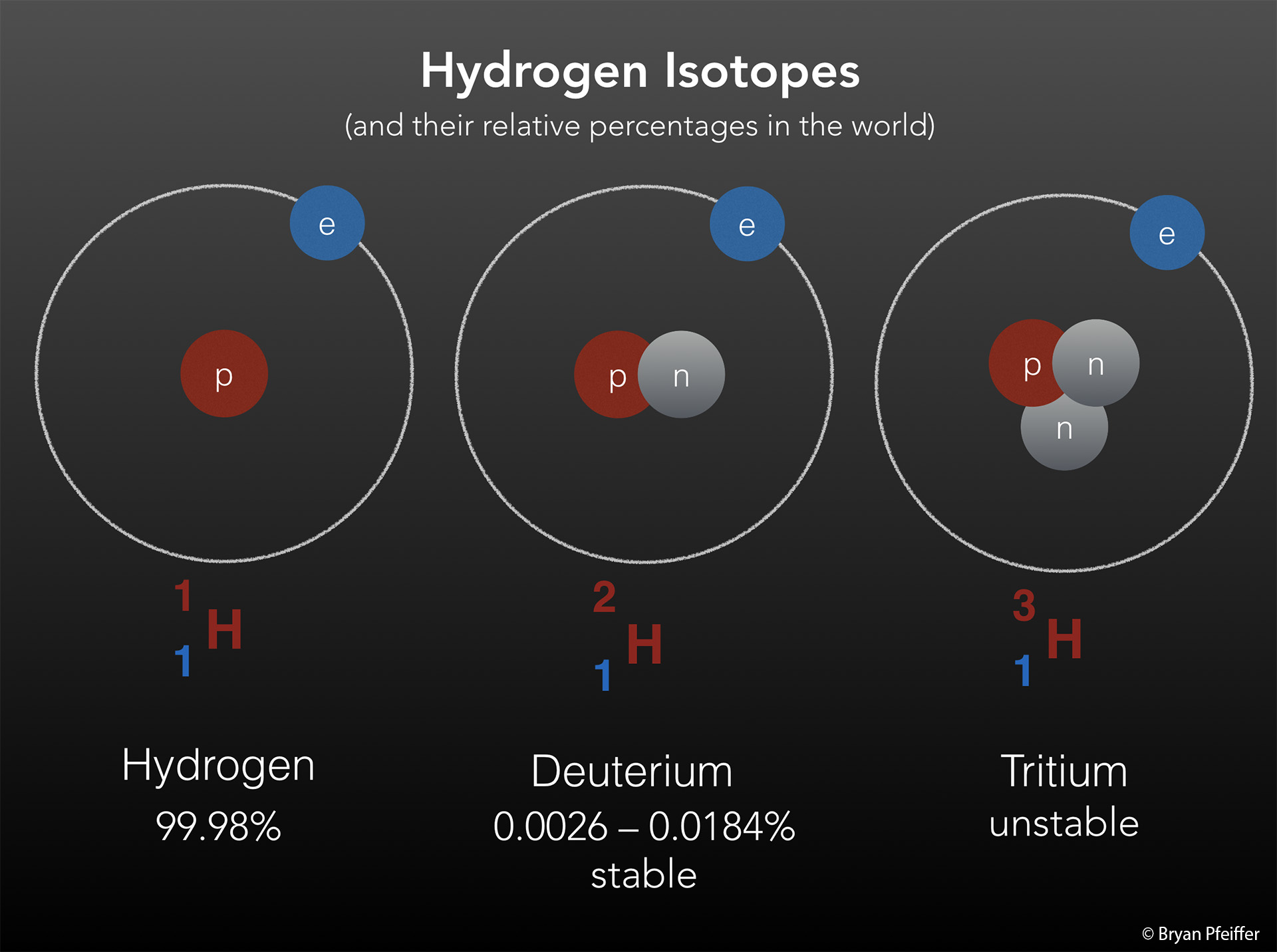 Recall that any atom has a nucleus, made up of protons and neutrons, surrounded by a cloud of electrons. An atom of the element carbon, for example, has a nucleus with six protons and six neutrons. It has an atomic mass of 12. And we call it Carbon 12. But nuclei don’t always play by the rules. Sometimes they might have extra or fewer neutrons. These variants are called “isotopes.” A carbon atom with an extra neutron (and yet still with its usual complement of six protons) has an atomic mass of 13. We call it Carbon 13. Lots of elements on the periodic table — hydrogen, oxygen, nitrogen and many others — can vary by way of these isotopes. They are always rare in the world. Some are unstable and decay (those are radioactive). But others persist in the environment for a long time. Those we call “stable isotopes.”
Recall that any atom has a nucleus, made up of protons and neutrons, surrounded by a cloud of electrons. An atom of the element carbon, for example, has a nucleus with six protons and six neutrons. It has an atomic mass of 12. And we call it Carbon 12. But nuclei don’t always play by the rules. Sometimes they might have extra or fewer neutrons. These variants are called “isotopes.” A carbon atom with an extra neutron (and yet still with its usual complement of six protons) has an atomic mass of 13. We call it Carbon 13. Lots of elements on the periodic table — hydrogen, oxygen, nitrogen and many others — can vary by way of these isotopes. They are always rare in the world. Some are unstable and decay (those are radioactive). But others persist in the environment for a long time. Those we call “stable isotopes.”
For biologists, even one extra neutron in a stable isotope can play an outsized role in wildlife research. Hydrogen turns out to be a fine element for tracking dragonflies. A hydrogen nucleus normally contains a single proton and no neutron. But a tiny fraction of hydrogen atoms around the world carry one proton and one neutron. We call that “heavy hydrogen,” or deuterium. It’s a stable isotope. (Tritium, with two extra neutrons, however, is unstable and radioactive.)
Stability has big benefits. The relative amount of deuterium in water, falling as rain or snow or just sitting there in a pond, is somewhat predictable around the world. It basically follows a concentration gradient corresponding with latitude. Water in Alberta, for example, carries a different deuterium-to-hydrogen ratio than water in Alabama. You can map it in the same way weather forecasters show us temperature gradients from north to south.
Because dragonflies grow up as nymphs in water, they incorporate that local deuterium-to-hydrogen ratio into their tissue. Wings and legs and most other insect body parts are metabolically inert — they form when the insect is “growing up” and do not change much when the animal is an adult. Adult insects, even when they eat, do not grow. So the deuterium-to-hydrogen ratio in a growing insect becomes kind of like a dialect that the adult carries throughout it high-flying life. So whether it happens to be flying in Costa Rica or Kenya, a dragonfly is unwittingly carrying around its signature deuterium ratio, its creation story. Deuterium is like a birth certificate that tells a biologist where a particular insect grew up. You are what you eat — or drink, as the case may be.
From this very collection, for example, my colleagues have traced the flight paths of another migrating dragonfly, Common Green Darner (Anax junius). And a biologist by the name of R. Charles Anderson (and colleagues) used these deuterium signatures to trace the flight of Pantala from India, across the Indian Ocean, with quick stop near his home on the Maldives, to destinations on the eastern shores of Africa — a flight of more than 6,000 kilometers, 3,500 of them over ocean. It turns out that all it takes to discover that flight path is an extra neutron in an atom of hydrogen.
And as if Pantala’s peregrination isn’t itself shocking enough, during my visits to the museum I wandered beyond the wanderer — toward the shock and awe of other insects neatly arranged on pins in wooden boxes with glass tops. Giant blue morpho butterflies from Brazil flashed like holograms beneath the collection’s florescent lighting. Beetles from Asia and the Americas glowed emerald green or displayed armaments that would make any self-respecting gladiator green with envy.
Among these vast collections of butterflies and beetles, dragonflies and bumblebees, I find order and symmetry, beauty and diversity. For me, embodied in any one dead insect is the force of history, science, discovery and awe.
Yet some of the world’s invertebrate collections have fallen into neglect (some have even been sent to landfills), and yet so much value and knowledge remains in the specimens. It’s as if Alfred Russel Wallace and other collectors did two centuries of field work for the rest of us. Our obligation now is to take these specimens and discover even more about how these animals live — or lived. The irony is that waiting for us on pins are species we have not yet fully discovered or understood — some of which are already extinct. Even those who find no such comfort among dead insects might instead appreciate something no less monumental that what it means to be human: the pursuit of knowledge. A tray of insects is like a great book, with individuals and drama yet to unfold.
Meet the Beetles
Consider here in Gainesville the impressive collection of Cerambycidae, the longhorn beetles, a group whose antennae are longer than their bodies. (For this beetle bliss we thank the late Edmund F. Giesbert, an expert on longhorns who left his spectacular collection to the FSCA.) One remarkable cerambycid here is Onychocerus albitarsis. No matter if you cannot pronounce it — just know that this beetle is so named in part for its white “feet” (tarsi). Yet this beetle’s acclaim is for its antennae.
Although they are the most diverse insect group on the planet, beetles never did manage to equip themselves with stingers. To be sure, many beetles defend themselves with toxins, which they exude from their bodies or, in the case of the bombardier beetles, spray out in a hot, nasty explosion out of their rear end. Because they lack fangs or a stinger to inject their toxins into their victims, beetles aren’t technically considered venomous. But Onychocerus is an exception — perhaps unique. It stings with the tip of its antennae. A beetle with antennae as weaponry — how cool is that? But it gets better. Onychocerus stings not like a bee or wasp. Its antenna delivers toxin like a genuine killer: a scorpion.
The business end of a scorpion, not technically a tail but rather modified terminal abdominal segments, is unmistakable — and visceral. There is little else like it in nature, certainly not among insects. But it turns out that Onychocerus’ stinger is almost identical to the scorpion’s — right down to tiny grooves or channels near the tip that help each arthropod deliver its toxin into the wound. Antenna are generally for love — not war. Beetles and other insects tend to use them instead for the senses — to locate plants for eating or pheromones for sex. We entomologists therefore don’t fear antennae.
An unsuspecting Herbert H. Smith got the point while picking up a cerambycid in Rio de Janeiro more than a century ago. “Seizing it with my forefinger and thumb, I was about transferring it to the collecting bottle, when, to my surprise, it inflicted on me a pretty sharp sting or prick, which caused me to drop it quickly,” Smith wrote in The American Naturalist (Smith 1884). “In defending itself thus, the insect used its antenna, spreading them out and then throwing them backward and upward with a strong jerk, so as to strike my finger and thumb just behind the nails with the scorpion-like terminal joint.”
Even so, the beetle and the scorpion do not share a common ancestor from which this injector was inherited. Their stingers evolved independently to be nearly identical. It appears to be a classic case of convergent evolution — unrelated organisms converging on the same form or function (in this case both) because it works in the struggle for existence.
More than a century after Smith’s surprising encounter with the beetle, biologists from New York and Peru confirmed the convergence with scanning electron microscopy, particularly those toxin grooves, which aren’t present in a similar non-venomous cerambycid beetle. And it turned out that the beetle was feisty in trying to defend itself from the biologist who had captured it. “Even after stinging the collector, O. albitarsis continued making attempts, suggesting that like scorpions, it can administer multiple stings,” the authors wrote (Berkov 2008).
So, here among 9 million dead insects we have a dragonfly that crosses oceans and a beetle that stings like a scorpion. And me wandering among them more than a century later, with a mere 8,999,998 more stories yet to be revealed.
References
Berkov A, Rodriquez N, Centeno P. 2008. Convergent evolution in the antennae of a cerambycid beetle, Onychocerus albitarsis, and the sting of a scorpion. Naturwissenschaften 95: 257–261
Smith, H H. 1884. Antennae of a beetle used as defensive weapons. American Naturalist 18(7): 727-728.







Well,Bryan…not sure “charmed” is the word..but must admit the Dogbane beetle is very colorful..(but cancel my tee shirt order !)
Well, Carol, give me a few minutes in summer, and I can almost guarantee that you’ll find some charismatic, lovely beetles. 🙂 They are, after all, the most diverse insect group. Google around for “Chrysina beetles” or even Dogbane Beetles. You’ll be charmed!
The butterflies are lovely but I cannot tolerate beatles …. it’s the worst “part” of summer for me. but I suppose the birds like them !!!
Aren’t the “stacks” amazing? I remember being among the echinoderm collection at the Smithsonian. Mind boggling! Just returned from 7 days of birding in Costa Rica and thought of you all the time.
Dragonflies everywhere
Most I’ve never met.
Dragonflies everywhere
and me without a net.
My best,
Judy
Nice piece
wow…that about covers it!
I can’t wait for the book either. It’ll be a while.
Hi Ruth,
Working title: Pantala: What an insect tells us about sex, evolution and the human condition.
B
It’s a wonderful mix of entomologists, some of whom are employed in the collection, plus visitors like me.
Hi Kathy,
The University of Florida has a huge natural history museum, specializing in Lepidoptera, the Maguire Center. That’s a wonderful public institution. http://www.flmnh.ufl.edu/mcguire/
The FSCA is indeed mostly set up for researchers.
Best,
Bryan
Can you visit the FSCA, or is it for research purposes only? Their website makes it look like you need to have ‘business’ there. thanks, K
Wow—all those cold case files. Do people have jobs here or are they are visiting researchers?
I’d still have my gaze enmeshed in the powdery wing scales (musical pun intended) of those iridescent be-bop-lepidops
so much buggery, so little time..
a wandering glider following the trail of the wandering glider. how lovely to pursue, just one of nature’s innumerable wonders. what will your book be called?
Very nice shots, and thanks for the summary of what you have been doing. The vortex has missed your presence!!
Wonderful! I can’t wait for the book!!! Thank you for the small yet engaging preview.